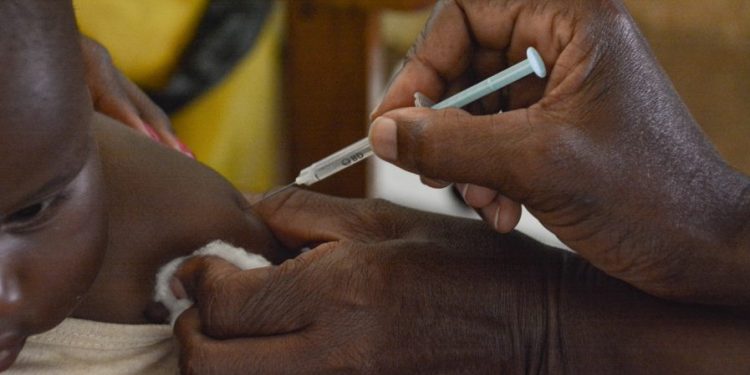
Ghana has approved the second malaria vaccine ever, paving way for the protection of more children aged five to 36 months.
One of the first countries to vaccinate against malaria through the pilot Malaria Vaccine Implementation Programme (MVIP), supported by the Vaccine Alliance (GAVI), Ghana has granted full national licensure to the R21/Matrix-M™ malaria vaccine, developed by the University of Oxford, United Kingdom (UK) and manufactured by the Serum Institute of India (SII).
Gavi stated yesterday that the second vaccine came alongside the RTS,S vaccine, which was prequalified by the World Health Organisation (WHO) in 2022, following a recommendation for its wider use in 2021.
According to the WHO’s 2022 world malaria report, the disease killed 619,000 patients in 2021 – with 96 per cent of those deaths in the WHO African region, making malaria one of Africa’s biggest killers. Alongside existing interventions, the vaccine is expected to help save more lives.
Gavi stated: “Ghana’s Food and Drugs Authority (FDA Ghana) has assessed trial data and approved the vaccine for use in children aged five to 36 months, the age group at highest risk of death from malaria. R21 has a three-dose primary series with a fourth (booster) dose a year later.
“R21’s dossier for pre-qualification has been accepted for review by the WHO, and we expect that Phase III results will be published soon. Should the WHO recommend the vaccine’s wider use, GAVI and United Nations Children Fund (UNICEF) could move to begin funding and procuring doses immediately upon the vaccine’s pre-qualification.
GAVI has already approved funding for a malaria vaccine programme and is ready to support adoption of R21 alongside RTS,S.”
GAVI also welcomed SII’s announcement of a technology transfer deal to produce the vaccine in Ghana, upon completion of a factory in Accra – which will complement GAVI’s own work with partners, including the African Union (AU) to support African vaccine manufacturing.
Meanwhile, the WHO endorsed another malaria vaccine, Mosquirix, developed by GlaxoSmithKline, last year. But lack of funding and commercial potential thwarted the production of enough doses, with just 15 million doses each year.
This is despite WHO saying roughly 100 million doses a year of the four-dose vaccine are needed long-term to cover around 25 million children.
Mid-stage data from the Oxford vaccine trial, involving more than 400 young children, was published in the Lancet in September 2022.
Vaccine effectiveness was 80 per cent in the group, which received a higher dose of the immune-boosting adjuvant component of the vaccine – an ingredient that helps create a stronger immune response in people receiving a jab.
Efficacy rates stood at closer to the 70 per cent mark in children in the lower-dose group, 12 months after their fourth dose.
The doses were administered ahead of the peak malaria season in Burkina Faso, West Africa. Data from an ongoing Phase III clinical trial in Burkina Faso, Kenya, Mali and Tanzania, that has enrolled 4,800 children, is expected to be published in a medical journal in the coming months.
Sim, a medical doctor and global expert in building healthy vaccine markets to ensure access in lower-income countries, said GAVI opened its first application window in July 2022 for support in rolling out the first ever malaria vaccine, to protect children against a disease that kills hundreds of thousands in Africa.
Sim added: “Two more application windows have opened since. GAVI, WHO and partners expect to announce allocations of the RTS,S vaccine soon.”
“This follows on from a decision by the Board of GAVI in December 2021 to approve an investment to support the malaria vaccine introduction, procurement and delivery for GAVI-eligible countries in Sub-Saharan Africa in 2022 to 2025.
“An initial investment of $155.7 million for 2022 to 2025 will initiate the implementation of this additional tool in the fight against malaria.” – Agency report.